Summary
By 2008, an estimated 1.46 billion adults globally were overweight (body mass index [BMI] =25 kg/m2) and 502 million adults were obese (BMI =30 kg/m2) [Finucane MM et al. Lancet 2011]. Furthermore, an estimated 170 million children (aged <18 years) globally were classified as overweight or obese; this estimate includes more than 25% of all children in some countries, more than double the proportions since the start of the epidemic [Lobstein T et al. Obes Rev 2004].
- Diabetes Mellitus
- Obesity
An overview of the global prevalence of diabetes was presented by Steven E. Kahn, MB, ChB, Veteran's Administration Puget Sound Health Care System and University of Washington, Seattle, Washington, USA. By 2008, an estimated 1.46 billion adults globally were overweight (body mass index [BMI] ≥25 kg/m2) and 502 million adults were obese (BMI ≥30 kg/m2) [Finucane MM et al. Lancet 2011]. Furthermore, an estimated 170 million children (aged <18 years) globally were classified as overweight or obese; this estimate includes more than 25% of all children in some countries, more than double the proportions since the start of the epidemic [Lobstein T et al. Obes Rev 2004].
Obesity is a prominent risk factor for type 2 diabetes mellitus (T2DM), and with global obesity rates rising, the ensuing burden of T2DM is also on the rise. By 2030, the number of individuals with diabetes worldwide is expected to rise to 435 million, almost 80% of whom will be in low- and middle-income countries [http://www.idf.org]. In the United States alone, 25.8 million people (8.3% of the population) now have diabetes [http://www.cdc.gov/diabetes].
Robert H. Eckel, MD, University of Colorado, Aurora, Colorado, USA, discussed the pathogenesis of obesity T2DM. According to Dr. Eckel, only 2% to 5% of severe human obesity is caused by rare monogenic mutations in the leptin central nervous system signaling pathway—likewise with monogenic syndromes of abnormal glucose homeostasis, such as Wolfram syndrome and Friedreich ataxia. While monogenic mutations account for <1% of T2DM, common gene variants account for 15%.
T2DM is characterized by insulin resistance and deficient β-cell function [Khan SE et al. Diabetologia 2003; Stumvoll M et al. Lancet 2005; Khan SE et al. Nature 2006]. To date, there are approximately 40 genes that have been associated with T2DM, most of which are β-cell genes.
Potential common underlying defects link obesity and T2DM. These factors include insulin resistance [Eckel RH et al. Lancet 2005], visceral fat distribution [Cnop M et al. Diabetes 2002], decreased β-cell mass [Butler AE et al. Diabetes 2003], increased adipokine secretion [Bashan N et al. Endocrinology 2007], a greater number of adipose tissue macrophages [Cinti S et al. J Lipid Res 2005], and β-cell decompensation [Weyer C et al. J Clin Invest 1999; Khan SE et al. Diabetologia 2003; Khan SE et al. Nature 2006].
Evidence suggests that there is a key role for the brain in controlling both body fat content and glucose metabolism. Neuronal systems that regulate energy intake, energy expenditure, and endogenous glucose production sense and respond to input from hormonal and nutrient-related signals that convey information regarding both body energy stores and current energy availability. In response to this input, adaptive changes promote energy homeostasis and the maintenance of blood glucose levels in the normal range. Defects in this control system are implicated in the link between obesity and T2DM [Schwartz MW et al. Science 2005].
Treatments and Treatment Interactions
Obesity is a potent risk factor for T2DM. Colditz et al. [Ann Intern Med 1995] found that BMI was the dominant predictor of risk for T2DM among 114,281 female registered nurses aged 30 to 55 years who were followed from 1976 to 1990. Compared with women with stable weight (gained or lost <5 kg between age 18 and 1976) and after adjustment for age and BMI at 18 years, the relative risk for diabetes mellitus among women who had a weight gain of 5.0 to 7.9 kg was 1.95 (95% CI, 1.5 to 2.3). The corresponding relative risk for those who gained 8.0 to 10.9 kg was 2.7 (95% CI, 2.1 to 3.3).
Common variants in the transcription factor 7-like 2 gene (TCF7L2) have also been associated with an increased risk of T2DM among people with impaired glucose tolerance. The risk-conferring genotypes in TCF7L2 are associated with impaired β-cell function but not insulin resistance. Regardless of genetic predisposition, lifestyle is a more prominent factor than genetic risk for diabetes [Florez JC et al. N Engl J Med 2006]. Weight loss surgery also produces metabolic improvement that is proportional to weight loss, with the greatest gains from biliopancreatic diversion and gastric bypass, followed by sleeve gastrectomy and gastric banding.
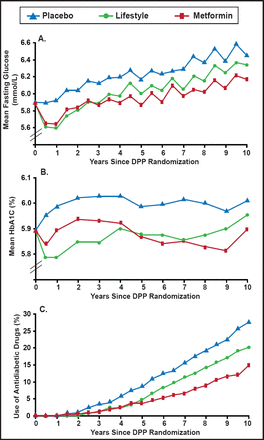
In the 2.8 years of the randomized Diabetes Prevention Program (DPP) clinical trial, diabetes incidence in high-risk adults was reduced by 58% with intensive lifestyle intervention and by 31% with metformin, compared with placebo [Knowler WC et al. N Engl J Med 2002]. During the 10-year follow-up, diabetes incidence in the placebo and metformin groups fell to that in the former lifestyle intervention group, but the cumulative incidence of diabetes remained lowest in the lifestyle intervention group (Figure 1), showing that delay of diabetes onset with lifestyle changes or metformin can persist for at least 10 years [Diabetes Prevention Program Research Group. Lancet 2009].
Fasting Glucose (A), HbA1C (B), and Antidiabetic Drug Use (C) Over Time Since the Initial DPP.
Reprinted from The Lancet. 10-year follow-up of diabetes incidence and weight loss in the Diabetes Prevention Program Outcomes Study. Diabetes Prevention Program Research Group. Copyright 2009. 374(9702): 1677–1686. With permission from Elsevier.
All participants were included, irrespective of whether they developed diabetes during follow-up. Study-assigned metformin was excluded from antidiabetic drug use.
Preferably, weight loss drugs should induce and sustain weight loss, improve cardiometabolic risk factors, and have acceptable adverse events and tolerability. Although there are numerous new targets and sites of biological action, the history of weight loss agents has been controversial at best [Eckel RH et al. J Clin Endocrinol Metab 2011. In press]. Despite improved understanding of obesity and numerous new targets, medications remain limited by toxicity, side effects, and lack of efficacy. Development of more effective, nontoxic weight loss medications remains an extremely high priority.
Obesity and diabetes share obvious features, but they are not one and the same; the genetic underpinnings of obesity differ from the genetic risk factors for hypoglycemia or T2DM. David M. Nathan, MD, Harvard Medical School, Boston, Massachusetts, USA, concluded that successful treatment of obesity prevents and ameliorates diabetes; behavioral modification provides more successful, long-term weight loss than most traditional “diets;” and it is critical to make effective therapies available.
Successful glycemic treatment is often associated with further weight gain, but some new medication classes appear to limit weight gain or even lead to weight loss. Whether these drugs are “worth” their added expense needs to be established. In the interim, the development of safe, effective antidiabetes drugs that do not cause weight gain is a high priority. Combining behavioral modification with weight loss medications can limit weight gain.
- © 2011 MD Conference Express®